Embryoid body differentiation from human iPSCs
Early embryogenesis is characterized by a tightly regulated sequence of lineage restrictions that ultimately leads to the formation of the three primary embryonic germ layers. Induced pluripotent stem cells (iPSCs) provide a powerful in vitro model to reproduce these developmental events.
When aggregated into three-dimensional structures known as embryoid bodies (EBs), iPSCs spontaneously differentiate and give rise to ectodermal, mesodermal, and endodermal derivatives.
EBs are therefore widely used as a model to study pluripotent stem cell differentiation and the early mechanisms of embryonic development. However, prolonged culture of EBs requires frequent medium changes and careful handling, which can introduce experimental variability.
Because automation is particularly relevant for sensitive cell cultures, and embryoid bodies represent both a delicate and widely used model, we evaluated the ability of Nestor, the automated cell culture system developed by Cellaven, to maintain and automate the differentiation of embryoid bodies derived from human iPSCs.
Automated embryoid body culture using Nestor

Nestor autonomously maintained the culture of human iPSCs (SCTi003-A, Stemcell Technologies) for two passages. As illustrated in Figure 1, once the cells reached the appropriate state, they were manually seeded into low-attachment U-bottom 96-well plates and cultured as self-aggregated spheroids for three days in eTeSR medium supplemented with blebbistatin (1:2000) (Stemcell Technologies).
Once fully formed, the spheroids were transferred to Matrigel-coated 24-well plates and supplied daily with fresh hESC medium by Nestor.
Over a period of 21 consecutive days, Nestor enabled continuous culture without manual intervention during weekends.
Embryoid body differentiation assessed by morphology and germ layer markers

Differentiating EBs were observed daily by brightfield microscopy to monitor matrix adhesion and cell spreading.
As illustrated in Figure 2, on day 1 EBs measured between 745 and 920 µm in diameter (right). By day 7 (center), cells had begun migrating and spreading from the original spheroid. By day 20 (left), the cultures had developed mature and organized structures displaying morphological features characteristic of early embryonic development.
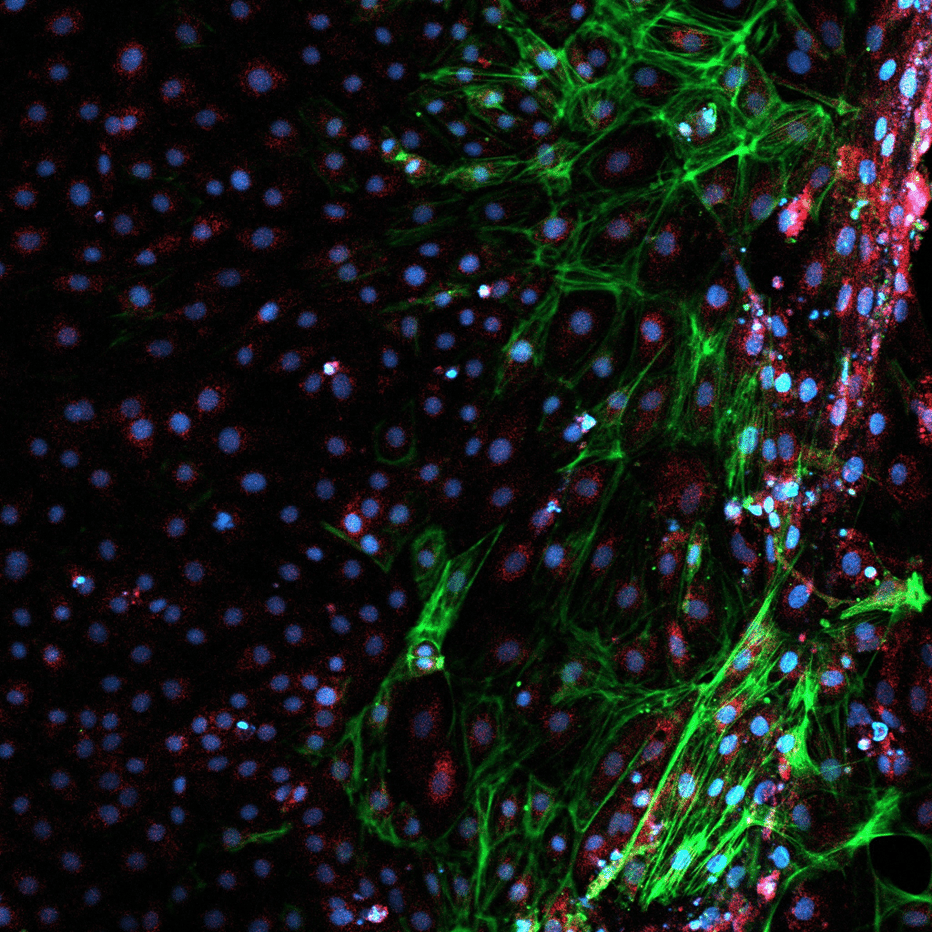
To determine whether the observed changes in gene expression translated into corresponding protein production, immunofluorescent staining of EBs was performed using antibodies targeting markers specific to the three embryonic germ layers: human α-fetoprotein (mouse, BioLegend), human α-SMA (rabbit, Abcam), and human β-tubulin III (mouse, BioLegend).
Due to cross-reactivity between some antibodies, endoderm and ectoderm staining were performed separately.
As illustrated in Figure 4, 21-day-old EBs express proteins characteristic of the three embryonic germ layers.
Stable embryoid body differentiation using Nestor
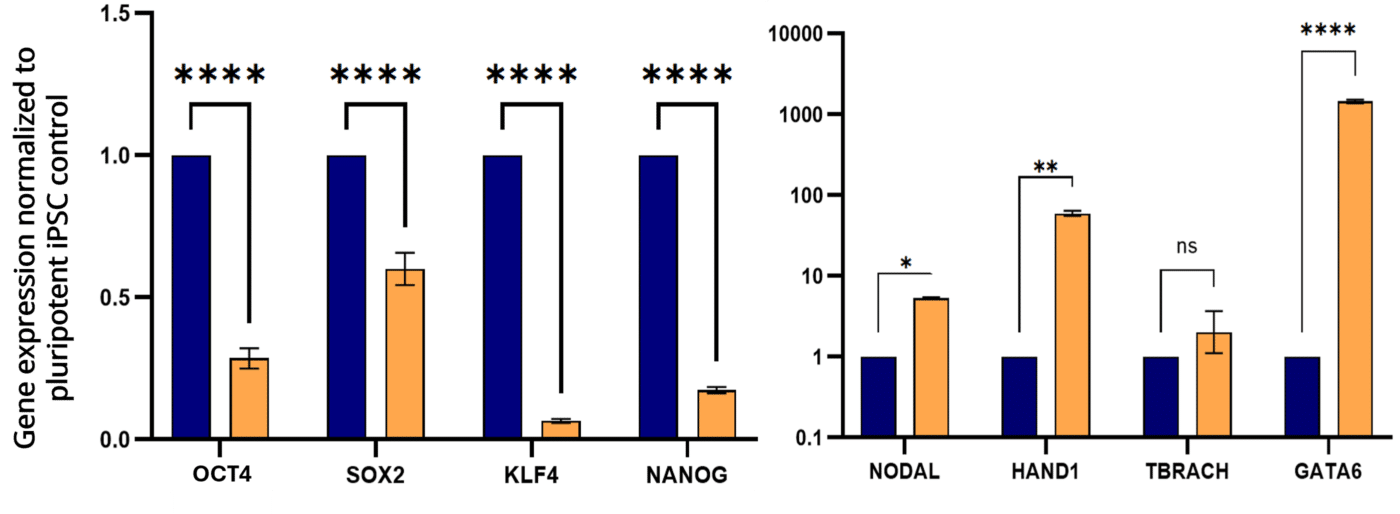
This study demonstrates Nestor’s ability to support prolonged differentiation of embryoid bodies. Automation did not alter the ability of iPSCs to spontaneously differentiate into derivatives of the three embryonic germ layers.
The gentle aspiration system integrated into the plate lid did not disrupt the morphology or adhesion of the embryoid bodies.
A stable culture environment allowed iPSC-derived spheroids to progressively lose pluripotency and express genes associated with the different embryonic germ layers.
At the functional level, automatically differentiated embryoid bodies also express key proteins in a spatially organized manner. This characteristic spatial organization suggests effective spontaneous differentiation and functional interactions between cells and their microenvironment.
References
- Pineda ET et al. Differentiation patterns of embryonic stem cells in two- versus three-dimensional culture. Cells Tissues Organs. 2013 Feb.
- Jaklin M et al. Focus on germ-layer markers: A human stem cell-based model for in vitro teratogenicity testing. Reproductive Toxicology. 2020.
Authors
Duncan Alric, Houda Djedaini, Harald Chaboud, Alexandre Houzé, Lisa Morisseau, Julien Maruotti
07/2025 – Cellaven, Besançon, France